Gather and Sow: April 2026

This month’s newsletter lands just in time for Easter. If you haven’t dyed eggs yet, consider making homemade dyes from a variety of plants. The recipe is below in Botany Lab of the Month: Dyeing Easter eggs, violet syrup, and learning about pH with plant pigments. Be sure that the plants you choose to make dyes with include beets and something tinted with purple anthocyanins (for example: red cabbage, dried hibiscus flowers, blueberries). Then you’ll be able to participate in the pH exploration portion of the activities. And if violets (genus Viola), either wild ones or garden pansies, are blooming already where you are, you can make violet syrup for even more pH exploration. That recipe is in this month’s Botany Lab of the Month, too. You’ll probably get more out of the lab exercises if you have already read this month’s Food for Thought: Betalains and the road not taken. Betalains are the pigments found in beets and their relatives. Plants that make betalains do not make anthocyanins. We tell the evolutionary tale of these two pigments.
The newsletter concludes with Gleanings and Baker’s Dozen, in which we highlight recent relevant media and journal articles. In the Baker’s Dozen of last month’s newsletter we highlighted a few foundational papers by female scientists, and we enjoyed that so much that we have decided to make a little retrospection a part of every newsletter’s literature round-up. The thirteenth entry to Baker’s Dozen will be an “oldie but goodie” significant paper.
Food for Thought
Betalains and the road not taken
Two roads diverged in a wood, and I—
I took the one less traveled by,
And that has made all the difference.
– excerpt from “The Road Not Taken”, by Robert Frost (1915)
A graduate student in 1918 was the first person to recognize that the red pigment in beets is wholly distinct from the classes of chemicals that cause red colors in most other plants (1–3). The young man, one G. Schudel, worked in the lab of Swiss chemist Richard Willstätter, the recipient of the 1915 Nobel Prize in Chemistry for his work on chlorophyll, the green plant pigment that harnesses light energy for photosynthesis. Schudel and Willstätter called the group of pigments “betacyanins.” The “beta” in the description is from the genus name for beets, Beta. And while “cyanin” ultimately derives from the Greek kyanós, meaning “blue,” Schudel and Willstätter were drawing a connection to anthocyanins, the abundant and diverse class of pigments responsible for reds, purples, and blues in most plant species. Their initial thought was that the beet pigments, which are now called betalains, were some weirdly nitrogenous category of anthocyanins. We now know that the metabolic pathways that lead to the two classes of pigments diverged long ago in evolutionary time, and the tale of these two pigments is an interesting story.

The betalainic plants, marching to their own beet
Betalains and anthocyanins are never found together in the same plant. Betalains wholly replace anthocyanins in plants that make them, from flowers to roots. Among plants, betalains are present only in a single large clade within the taxonomic order Caryophyllales (the Core Caryophyllales). Betalains also independently evolved in some basidiomycete fungi, including the charismatic fly agaric mushroom (Amanita muscaria), and a few bacteria. With a few exceptions (see “There and back again” and the phylogeny figure below), all the species in the Core Caryophyllales produce betalains and not anthocyanins (4–6).
The Core Caryophyllales is home to around 9000 species divided among 29 taxonomic families. From a human perspective, the Amaranthaceae may be the most important betalainic family. It is where we find beets and chard (both Beta vulgaris), spinach, amaranth (the edible varieties grown for grain and greens, as well as the colorful amaranths grown as garden flowers), quinoa, and sea beans (Salicornia). All cactuses (Cactaceae) are betalainic, including the bright magenta and gold-colored fruits of prickly pear (Opuntia spp.) and dragonfruit (Selenicereus spp.). Malabar spinach (Basella spp; Basellaceae) is a unique vegetable that is reminiscent of a vining purslane (Portulaca; Portulacaceae), both of which are betalainic. And betalains are responsible for the brilliant floral whorls of ice plants, four o’clocks, and bougainvilleas (all from the Nyctaginaceae).
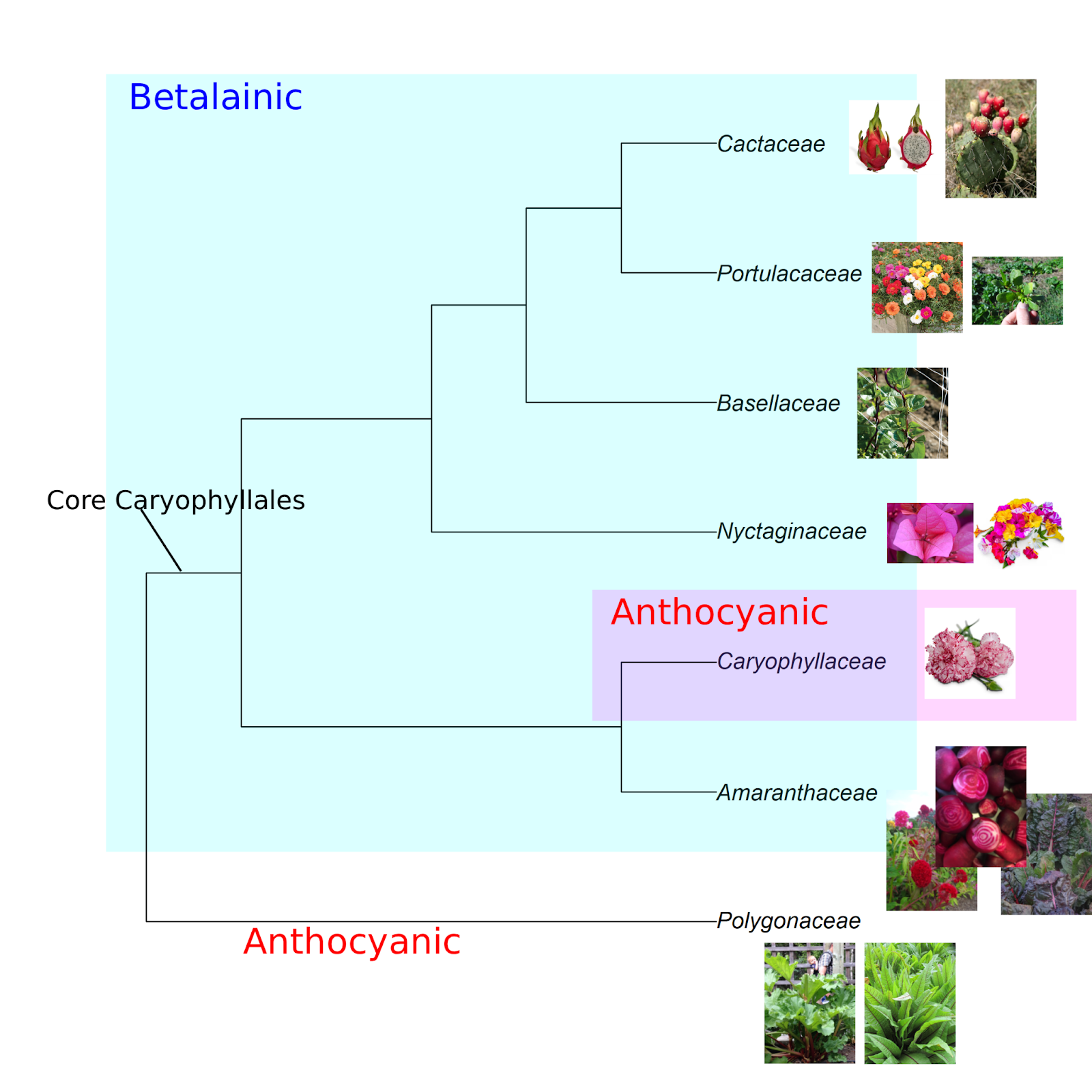
Nature’s paintbrushes
Outside the Core Caryophyllales, anthocyanins are ubiquitous across almost all land plants.The gorgeous reds in rhubarb (Rheum × hybridum) are anthocyanins. Rhubarb’s taxonomic family, Polygonaceae (the buckwheat family), is in the Caryophyllales, but it is evolutionarily basal to (diverged earlier than) the Core Caryophyllales and is therefore anthocyanic and not betalainic (see the phylogeny figure above).

Anthocyanins have the color market cornered when it comes to true blues, but, otherwise, there is remarkable overlap in the purple-to-red palettes produced by anthocyanins and betalains. High concentrations of either can even produce black (see the ripe betalainic pokeberries and anthocyanic blackberries in the figure below). Similarly, the yellow-gold betalains, on display in golden beets, are virtually indistinguishable from colors produced by carotenoids, which is another major category of plant pigments. Carotenoid colors range from yellow (as in sweet corn), to orange (as in carrots) and red (the red in tomatoes and watermelon is from the carotenoid lycopene). Unlike anthocyanins, carotenoids and betalains are not mutually exclusive. As we discuss below, plant pigments are physiologically important, not just colorful, and while betalains are able, more or less, to perform the same functions as anthocyanins (7), there are some jobs that only carotenoids can do (8).

More than just a pretty face
Why do plants make pigments? The first thing that comes to mind is that color helps flowers and fruits attract helpful animals, for pollination or seed dispersal, respectively (9). Beyond animal attraction, pigments perform numerous physiological functions (2, 7, 8, 10–14). Betalains and anthocyanins act as sunscreens, to absorb and block excess light and ultraviolet radiation, and as antioxidants, to repair damage caused by what they can’t block. Drought, cold, and salinity stress also induce plants to produce pigments. These additional environmental stressors can make plants more susceptible to harm by excess light in addition to introducing their own unique difficulties. Plant pigments help plants rise to the challenge. Both anthocyanins and betalains have also been shown to protect plants from fungal or microbial pathogen attack. Pigments are also involved in mediating interactions between plants and beneficial microbial symbionts (15).
Plant pigments can also serve as antioxidants in animals–people included–that eat the plants. There is strong evidence that some animals preferentially consume colorful fruit not because it is easier to see but because it is a good source for antioxidants (1, 16).
Two (metabolic) roads diverged in a yellow wood (or, more likely, in some inhospitable treeless steppe)
It remains something of a mystery as to why betalainic plants never make anthocyanins as well (3–6, 17–23). All of the genetic machinery for making anthocyanins is still present in betalainic plants, and betalainic plants still make other flavonoids, which is the larger class of protective chemicals that includes anthocyanins. The gene regulatory mechanisms that act as on/off switches to the anthocyanin biosynthesis pathway simply never get turned on. There is not a definitive answer to why this is the case, but we do know that there is an inherent competition for construction materials between the two pigment types. Anthocyanins and betalains are both made from the same initial molecule (arogenate), and species appear to commit fully to producing one or the other.
Arogenate is the precursor molecule for two amino acids: phenylalanine and tyrosine. Phenylalanine is the first molecule in the anthocyanin pathway, and tyrosine is the first compound in the betalain pathway. Arogenate is transformed into tyrosine by the enzyme ADH. After that, the betalain synthesis pathway comprises three main steps, each performed by a specific enzyme, ending with the production of betalamic acid. Betalamic acid can then be converted to either the red-violet betacyanins (the name that Schudel and Willstätter originally used now has this more restricted definition), or the yellow-gold betaxanthins. Red table beets produce an abundance of betacyanins, while golden beets are tinted with betaxanthins.

The evolution of the betalain pathway was made possible by two key genetic accidents that resulted in the buildup of huge volumes of tyrosine inside plant cells. Before this series of events, and in flowering plants outside of the Core Caryophyllales, there was only one copy of the gene that coded for ADH, the enzyme that turns arogenate into tyrosine. This original version of the enzyme came with a built-in regulatory mechanism that strongly limited the amount of tyrosine produced. Plants need tyrosine for myriad functions, but not that much of it, and the original ADH ensured that most of the available arogenate could be converted into phenylalanine, much of which became anthocyanins.
Then, a gene duplication event (our first accident) happened in one of the ancestors of the Core Caryophyllales. Gene duplication events are mistakes during gamete production that leave an extra copy of genetic material in gametes. The size of the extra material can be as small as a single nucleotide (the individual building blocks of DNA) or as big as the entire genome (all the chromosomes). This sort of thing is common in plants and is enormously consequential (24, 25) because the extra copies of genetic material evolve independently. The original operational version of the genetic material keeps the lights on, so to speak, leaving the extra copy free to do fun things. Evolutionary changes to the copies can lead to remarkable innovations, strongly contributing to the diversity of plant species, form, and function.
Such was the case with the extra copy of ADH, called ADH-alpha. Shortly after its appearance, ADH-alpha relaxed its tight regulation of tyrosine production (the second of our two genetic mishaps). It gobbled up the available arogenate and left a glut of tyrosine in its wake. This left the anthocyanin pathway pressed for raw materials and the evolutionary field open for further innovations to deal with both the excess tyrosine and the need for pigments.
The enzymes that transform tyrosine into betalamic acid were all originally part of other physiological processes and were co-opted into the secondary function of dealing with the piles of tyrosine. This sort of thing—molecules serving more than one function or finding themselves surprisingly able to do a new task—is also common in plant evolution.
There and back again
Some formerly betalainic plants within the Core Caryophyllales have reverted to anthocyanin production and have ceased making betalains (26, 27). This reversion has happened independently multiple times in a handful of families (the Caryophyllaceae, home to carnation flowers (Dianthus spp.), are one of these). In every case, genetic mutation has damaged the ADH-alpha that allowed for the buildup of tyrosine. With tyrosine production curtailed, the anthocyanin pathway was able to resume, millions of years after it was initially turned off. This was probably only possible because the genetic architecture for other flavonoids was still actively used by betalainic plants and therefore not subject to the evolutionary version of decay, rust, and ruin.
Plant pigments in the kitchen

Anthocyanins and betalains are similar in many ways, but they respond differently to temperature and their chemical environment (pH and salinity, among other things). This has evolutionary implications (7, 28), but also culinary ones. As the title to Samin Nosrat’s cookbook and Netflix show so aptly summarizes–Salt Fat Acid Heat–temperature, pH, and salinity are central aspects of food preparation. The color of anthocyanic and betalainic foods can change depending on what process you put them through. In general, the color of betalains tends to be more stable than that of anthocyanins. We will see this firsthand in this month’s Botany Lab of the Month exercise, below.
Botany Lab of the Month
Dyeing Easter eggs, violet syrup, and learning about pH with plant pigments
Dyeing Easter eggs with homemade edible plant dyes makes for some superb kitchen botany. Making the dyes is easy, fun, and offers insight into the diversity and chemistry of plant pigments. Nature’s ancient art supplies enhance the color and nutrition of our food. The dye plants we will talk about here are pictured in the gorgeous illustration by The Kitchn, from the blog post about their experiences using plants to dye eggs.

The pigments in this particular set of plants give us the opportunity to turn our egg dyeing project into a laboratory about pH indicators and the sensitivity of different plant pigments to pH. The pH scale is used to define a solution’s acidity or alkalinity. It is a logarithmic scale, from 0 to 14, that represents the concentration of hydrogen ions (H+). A pH value of 7, in the middle, is neutral. Pure water is neutral. Values lower than 7 are acidic. Values higher than 7 are basic.
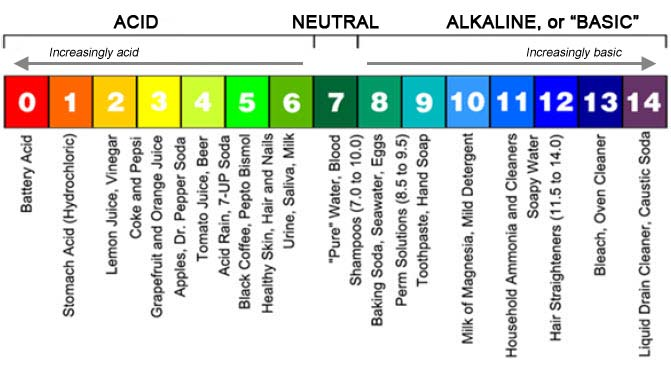
The original version of the plant pigment indicator experiment was conducted in the 1660s by one of the fathers of modern chemistry, British scientist Sir Robert Boyle. He used a syrup made from wild violets as his pH indicator, although the pH scale had not been invented yet. Making lemony and non-lemony versions of beverages with violet syrup offers another opportunity to explore the pH sensitivity of anthocyanin plant pigments. We include the violet syrup recipe, and a little bit more about Boyle, after the egg dyeing recipe.
Which pigments are we dealing with here?
Compare the colors of the plant material to the colors of the dyed eggs in the photo. The eggs dyed with red cabbage and hibiscus are noticeably more blue than the reddish-purple plant tissue (leaves and sepals, respectively). The same is true to a lesser degree with blueberries. This discrepancy is the result of the pH-sensitivity of anthocyanins, the dominant pigments in those plants. Plants have ways to deal with this to maintain a particular color, such as pairing anthocyanins with other molecules (co-pigments, usually other flavonoids), or making special storage compartments (vacuoles) for them inside plant cells (29–33). We will further explore pigments and pH in the egg dyeing recipe section below, and in the violet syrup section that follows.

Onion skins also have anthocyanins, particularly in red onions, but they also have high concentrations of other flavonoid pigments that modulate the color swings and contribute the ochre color in the onion skin-dyed eggs (34). Beets, as we described above in Food for thought: Betalains and the road less traveled, owe their vibrant colors to betalains, not anthocyanins. Betalains are far more pH-stable than anthocyanins, which is why the beet-dyed eggs more closely match the source material. The same is true for the curcumoinoids responsible for turmeric’s golden color and distinctive earthy flavor (35).
All of the pigments in play here are phenolics, the enormous set of compounds whose synthesis begins with one of the three amino acids manufactured in the plant Shikimate pathway: tyrosine, phenylalanine, and tryptophan (36–38). Betalain synthesis starts with tyrosine, while anthocyanins start with phenylalanine, along with all of the rest of the flavonoids (important in onion) and other phenylpropanoids (in turmeric).

Carotenoids, another major class of plant pigments that we mentioned in the Food for Thought essay, are not phenolics. While all plants make at least some carotenoids, which serve essential physiological functions, carotenoids do not appreciably contribute to the colors of the plants on our list. The dye recipe (below) is generic, though, so feel free to try it with carrots or tomatoes to explore the egg dyeing capacity of some carotenoids. Or try parsley, for that matter, to experiment with the green pigment chlorophyll (also not a phenolic). Incidentally, the yellow-orange color of the yolks inside the dyed eggs is created by carotenoids that the chickens obtained from their food, ultimately from plant sources.
The egg dye recipe and pH indicator experiment

You’ll need to hard boil eggs before you dye them.
The basic recipe for all the edible plant dyes is the same: Combine 2 cups of coarsely chopped plant material (or 1 tablespoon turmeric powder) and 4 cups water in a saucepan, bring to a boil, then turn the heat down and simmer for 15-30 minutes. You don’t have to strain out the solids, but you can if you want to.
Reserve a few tablespoons of the strained liquid from simmered beets and something anthocyanic (cabbage, hibiscus, blueberries)--keep them separate.
To the main dye bath, add an edible acid (1 tablespoon white vinegar, or a half teaspoon of citric acid powder if you don’t want the vinegar smell). Add the hard-boiled eggs to the acidified dye bath and let it sit for 12-24 hours. Alternatively, experiment with repeated dips in the dye bath for shorter periods, instead of a single long soak. Let the eggs air dry in between dye dips.
For your beet and anthocyanic plant dye baths, compare the color of the reserved liquid (with no acid in it) to the color in the dye bath (with acid). Adding acid should not have changed the color of the beet liquid. The anthocyanic dye baths, however, should be noticeably more red after acid addition. The non-acidified extract from the anthocyanic plants is a pH indicator, meaning its color changes in a predictable way in response to the acidity or alkalinity of the solution that is added to it. You can explore the impact of adding an alkaline solution, too. Put a small amount of the indicator liquid (like, a teaspoon or two) in a separate dish. Add a pinch of baking soda (sodium bicarbonate) and stir. The liquid should turn grayish-blue. The eggs dyed in an anthocyanic bath are bluer than the indicator liquid because the calcium-based compounds in the eggshells are alkaline.




The two glasses in the center image contain a small amount of juice from pomegranate (left glass; pomegranate pictured at left) and beets (right glass; beets pictured at right). Baking soda (sodium bicarbonate) was stirred into both juices before this photo was taken. Before the addition of baking soda, the pomegranate juice was red, looking very similar to the juicy seeds in the ripe fruit, and also very much like beet juice. The red in pomegranate is from anthocyanin pigments, the color of which is sensitive to pH. Anthocyanins turn grayish-blue or green when exposed to alkaline solutions, like baking soda. Beets are colored with betalain pigments, the color of which is far more stable across a wide range of pH. Photos by J. Osnas and K. Preston.
Violets are blue…unless you add lemon juice to them
Changing the color of an anthocyanic liquid by adding acidic or basic solutions seems pedestrian to us now, but it was intellectually revolutionary in the 17th century. It was the motivating exercise described in Robert Boyle’s 1664 treatise Experimental History of Colours (39). He demonstrated that not only could you change colors through chemistry alone, as opposed to simply mixing different colors together, but that the color change could be diagnostic of the characteristics of the substance added to the anthocyanin. Boyle showed that the acidity or alkalinity of a solution could be inferred by adding it to violet syrup and observing its impact on the color, although he didn’t use the words “acidity” or “alkalinity.” He did, however, include a recipe for violet syrup!

The edible flowers of violets (Viola spp) are mild, pleasant, and beautifully, well, violet. Chef Kaity Farrell shares several ways to use them on her website (40). When imported granulated sugar became widely available in the 16th-17th centuries, one of the first things Europeans did with it was to make violet syrup, a popular sweet and medicinal treat (41). The recipe is simple. Make a strong infusion, or “tea”, by steeping the flowers in simmering water. Then, strain out the flowers and dissolve an equal volume of sugar into the hot liquid.
Kaity recommends making blue-purple beverages with the syrup, and then dazzling your friends by adding drops of lemon juice to their drinks at the table. The acid in the lemon juice tilts the color towards red. You could tilt the color toward green by adding something chemically basic, like baking soda (sodium bicarbonate), washing soda (also called soda ash, sodium carbonate), or pickling lime (calcium hydroxide). Unlike the lemon juice drops, however, the alkaline additions would yield an unpalatable result.


Violet syrup made by chef Kaity Farrell before (left) and after (right) the addition of lemon juice (40).
One of the modern ways to measure pH is with litmus paper indicator strips (such as these), which are imbued with a chemical–the indicator–that responds to acidity and alkalinity with particular color changes. You dip the strip into a solution and compare it to a color key to diagnose pH. The strips are just a more repeatable version of adding stuff to violet syrup–or red cabbage juice–and seeing what happens.
Across its nearly 700 species, the genus Viola has a widespread distribution in the Northern Hemisphere. Spring has sprung across much of its range. Perhaps violet lemonade will accompany some plant-dyed Easter eggs this Sunday.
Gleanings and Baker's Dozen
Gleanings
- We are looking forward to reading How Flowers Made Our World: The Story of Nature’s Revolutionaries by David George Haskell. Interviews with the author about this work are universally worthwhile.
- Viviane Callier’s interview with Haskell in Nautilus is delightful.
- Cassandra Quave’s interview with Haskell on her Foodie Pharmacology podcast
- The 2026 National Geographic 33 “visionary changemakers” includes Italian botanist Benedetta Gori, a champion of edible wild plants.
- People love to travel just to marvel at amazing floral displays. The New York Times has an article about using AI to predict peak cherry blossom time in Japan. The Washington Post predicted that D.C. cherry trees would be late this year, peaking this week. They even include data. But you don’t have to take their word for it. The web cam says it’s already happening. And before the cherries bloom in Seoul, Korea, the forsythia come out, turning parts of the city a cheery yellow. We just missed that festival, but maybe next year.
- And of course both Arbor Day and Earth Day happen in April, providing loads of opportunities to celebrate plants in your life.
Baker’s Dozen
In last month’s newsletter we highlighted a few foundational papers by female scientists, and we enjoyed that so much that we have decided to make a little retrospection a part of every newsletter’s literature round-up. The thirteenth entry to Baker’s Dozen will be an “oldie but goodie” significant paper.
- Fujita, T., et al. (2025). The flower does not open in the city: evolution of plant reproductive traits of Portulaca oleracea in urban populations. Annals of Botany, 135(1-2), 269-276. Ever wonder why you never see flowers on the purslane that grows in random urban patches of dirt? This crunchy juicy edible weed makes two kinds of flowers: those open to pollinators (“chasmogamous”) and those that self-fertilize inside a never-opening bud (“cleistogamous”). Many species make both kinds on the same plant, but individual purslane plants are genetically programmed to make only one or the other kind. This paper documents natural selection favoring closed-bud cleistogamous genotypes in cities in Japan.
- Macías-Naranjo, S. M., et al. (2026) Amaranth, the Ancient Pseudocereal: A Promising Crop for Climate-Resilient Agriculture and Healthy Diets. Frontiers in Plant Science, 17, 1716624. A very comprehensive review of everything you ever wanted to know about amaranth, its past, present, and future. Amaranth is an old crop with edible seeds and leaves, great standing genetic diversity, and the potential to withstand a lot of abiotic stress. The authors explore whether it could be a new supercrop. Amaranths can be very colorful–and all those reds and golds are from betalains (see this month’s Food for Thought essay above).
- Belvisi, G., & Tranchina, G. Projected shifts in climatic suitability of olive (Olea europaea L.) in the Mediterranean. Frontiers in Plant Science, 17, 1743577. Where will olive trees thrive in the future? This study models the suitability of areas around the Mediterranean Basin to support olive growth as temperatures rise. Warmer nights in southern France will make that region more hospitable for the trees, while heat stress may limit tree growth in some currently cultivated areas. The authors caution that other factors, biological and social, may limit the success of olives in their new homes.
- Prusky, D. B., et al. (2026). Quiescence of postharvest pathogens: a fungal inhibition process or an immune response of the unripe host fruit?. New Phytologist. Do you ever look at the tomatoes or bananas or peaches ripening slowly on your countertop and see them as living beings, dying slowly, and under siege by a community of microbes waiting patiently for them to surrender? This paper tells their story.
- Noraz, R., et al. (2026) Ancient DNA reveals 4000 years of grapevine diversity, viticulture and clonal propagation in France. Nat Commun 17, 2494 (2026). Grapevine cultivation has never been a casual affair in France, as this study shows. Genotyping of grape seeds from archeological sites that span several millenia reveal long-distance exchange of domesticated genotypes and the careful clonal propagation of favored varieties, especially pinot noir.
- Scossa, F., et al. (2026) Convergence and parallelism in the evolution of plant metabolism. JIPB preprint. A great review about a topic essential to understanding the evolution of our food plants
- Wojcik, C., et al. (2026) Evolution of crop phenotypic spaces through domestication. New Phytologist preprint. An admirable entry in the field of quantifying how domestication has changed plant phenotypes.
- Alvey, E., et al. (2026) A manifesto for plant science education. Plants People Planet 8: 422-432. A persuasive argument and priorities for more plant science education at all levels.
- Two regional perspectives on nutritionally significant wild plant foods
- Huang, L., et al. (2026) Wild vegetables contribute to improving the diversity and sustainability of vegetable diets in China. PNAS 123: e2524063123
- Abdellatef, E., et al. (2026) Prospective of indigenous African wild food plants in alleviation of the severe iron deficiency anaemia in Sub-Saharan Africa. Plants People Planet 8: 486-498.
- Engelgau, P., et al. (2026) Banana aroma is a result of acetohydroxyacid synthase and isopropylmalate synthase alternative isoforms that bypass feedback inhibition. PNAS 123: e2515301123. A definitive explanation of banana aroma and why it is otherwise rare in nature.
- Bolei Chen, Y. Z., et al. (2026) Foliar dewdroplet–induced redox cascades promote early flowering in Brassicaceae plants. PNAS 123: e2527021123. Dewdrops are a heretofore underappreciated environmental trigger for plant developmental stages. Let the poetry commence.
- The journal formerly called Economic Botany is now called Ethnobotany and Economic Botany, accompanying the name change of the Society for Economic Botany to the Society for Ethnobotany. Some recent papers from the journal that are particularly relevant to our purposes include:
- Urban, M. (2026) Indigenous Nomenclature Reveals Prehistoric Interaction Related to Cacao in Amazonia and Diffusion to Mesoamerica. Ethnobotany and Economic Botany preprint. Linguistic clues about the identity of the particular strain of Theobroma cacao to escape Amazonia and the pattern of its human-mediated dispersal across the Americas four millennia ago.
- Bourdeix, R., et al. (2026) Controlled Pollination and Reproductive Strategies in Coconut: A Framework for Farmer-Led Breeding, Seednut Production, and In Situ Conservation. Ethnobotany and Economic Botany preprint. Coconut farming is surprisingly finicky. This paper is complementary with another recent coconut paper from the journal this past fall: Gunasekara et al. (2025), linked below.
- Yessoufou, K., et al. (2025) Random Patterns of Medicinal Plants on a Phylogeny do not Imply Random Selections of Medicinal Plants. Economic Botany 79: 310-323. This paper deals specifically with medicinal plants, but medicinal plants and food plants exist on a continuum. We think a lot about what the phylogenetic pattern of food plants means for how we should understand the evolution of our diets. This paper is a useful contribution to that line of inquiry.
- Gunasekara, I. K. S., et al. (2025) Identification and Preservation of the Hawaiian “Niu Hiwa” Within Hawai‘i’s Coconut Diversity. Economic Botany 79: 360-379. There are a lot of different types of coconuts. Preserving genetic resources from the varieties is difficult.
- Wagner, J. G., et al. (2025) Exploring the Sociocultural Importance of Butia eriospatha (Arecaceae), an Iconic Palm of the Highlands of Southern Brazil. Economic Botany 79: 342-359. Edible and other uses of a palm species that survives in highlands that freeze every winter. It’s fun to learn about frost-tolerant palms–with edible fruit and seeds, no less.
- Oldie but Goodie: Ehrlich, P. R., and P. H. Raven (1964) Butterflies and plants: a study in coevolution. Evolution 18: 586-608. Paul Ehrlich passed away a few weeks ago (please refer to the press release from the Stanford School of Humanities and Sciences, and the obituary in Nature). He was an extraordinary biologist and a generous mentor at Stanford. One of his foundational contributions was the paper he wrote with botanist Peter Raven in 1964 that launched the modern study of coevolution. The notion that interacting species can reciprocally affect the evolution of each dates back arguably to Charles Darwin. Upon examining the extraordinarily long nectar spur on the Malagasi orchid Angraecum sesquipedale, Darwin famously posited that there must be a moth with a proboscis capable of plumbing it. The insect in question would not be identified until decades after his death (the description of the orchid on the Kew Gardens website includes a lovely video explanation). What Ehrlich and Raven accomplished with their 1964 paper was to establish criteria for determining when coevolution has occurred, and they documented coevolution between numerous butterfly clades and the defensive chemistry of their hostplants. The chemical evolutionary arms race between plants and the insects that wish to eat them demonstrably pushed not only the evolution of the relevant respective metabolisms but the speciation and diversification of both plants and insects. We talk a little about this in our old blog post about the effect of caterpillars on the chemistry of the cruciferous vegetables in the garden, one of the plant-butterfly pairs included in Ehrlich and Raven (1964). There have been a few review papers over the decades that have revisited Ehrlich and Raven (1964) and reaffirm that coevolution is a significant aspect of plant and animal evolution. In particular we recommend:
- Janz, N. (2011) Ehrlich and Raven revisited: mechanisms underlying codiversification of plants and enemies. Annual Review of Ecology, Evolution, and Systematics 42: 71-89.
- Rodrigo, C., et al. (2022) Ehrlich and Raven escape and radiate coevolution hypothesis at different levels of organization: Past and future perspectives. Evolution 76: 1108-1123.
References
1. G. J. Kapadia, G. S. Rao, “Anticancer Effects of Red Beet Pigments” in Red Beet Biotechnology: Food and Pharmaceutical Applications, B. Neelwarne, Ed. (Springer US, Boston, MA, 2012; https://doi.org/10.1007/978-1-4614-3458-0_7), pp. 125–154.
2. E. L. Bastos, W. Schliemann, “Betalains as Antioxidants” in Plant Antioxidants and Health (Springer, Cham, 2021; https://link.springer.com/rwe/10.1007/978-3-030-45299-5_9-2), pp. 1–44.
3. G. D. Moghe, S. D. Smith, The push and pull of plant specialized metabolism underlies a long-standing, colorful mystery. New Phytol. 217, 471–473 (2018).
4. V. E. Tossi, L. Martínez Tosar, S. I. Pitta-Álvarez, H. F. Causin, Casting light on the pathway to betalain biosynthesis: A review. Environ. Exp. Bot. 186, 104464 (2021).
5. H. Sheehan, T. Feng, N. Walker-Hale, S. Lopez-Nieves, B. Pucker, R. Guo, W. C. Yim, R. Badgami, A. Timoneda, L. Zhao, H. Tiley, D. Copetti, M. J. Sanderson, J. C. Cushman, M. J. Moore, S. A. Smith, S. F. Brockington, Evolution of l-DOPA 4,5-dioxygenase activity allows for recurrent specialisation to betalain pigmentation in Caryophyllales. New Phytol. 227, 914–929 (2020).
6. F. Gandía-Herrero, F. García-Carmona, The dawn of betalains. New Phytol. 227, 664–666 (2020).
7. G. Jain, K. S. Gould, Are betalain pigments the functional homologues of anthocyanins in plants? Environ. Exp. Bot. 119, 48–53 (2015).
8. H. Hu, N. Pradhan, J. Xiao, R. Xia, P. Liao, Chromatic symphony of fleshy fruits: functions, biosynthesis and metabolic engineering of bioactive compounds. Mol. Hortic. 5, 19 (2025).
9. M. Sinnott-Armstrong, L. Maier, S. D. Smith, A. S. Dellinger, Flower clades and fruit clades: Trade-offs in color diversification across angiosperms. Am. J. Bot. 113, e70146 (2026).
10. N. Grünig, J. M. Horz, B. Pucker, Diversity and ecological functions of anthocyanins. BMC Plant Biol. 26, 146 (2025).
11. K. M. Davies, M. Landi, J. W. van Klink, K. E. Schwinn, D. A. Brummell, N. W. Albert, D. Chagné, R. Jibran, S. Kulshrestha, Y. Zhou, J. L. Bowman, Evolution and function of red pigmentation in land plants. Ann. Bot. 130, 613–636 (2022).
12. H. A. Maeda, A. R. Fernie, Evolutionary History of Plant Metabolism. Annu. Rev. Plant Biol. 72, 185–216 (2021).
13. G. Li, X. Meng, M. Zhu, Z. Li, Research progress of betalain in response to adverse stresses and evolutionary relationship compared with anthocyanin. Molecules 24 (2019).
14. R. V. Espley, L. Jaakola, The role of environmental stress in fruit pigmentation. Plant Cell Environ. 46, 3663–3679 (2023).
15. I. A. Stringlis, R. de Jonge, C. M. J. Pieterse, The Age of Coumarins in Plant–Microbe Interactions. Plant Cell Physiol. 60, 1405–1419 (2019).
16. H. M. Schaefer, Why fruits go to the dark side. Acta Oecologica 37, 604–610 (2011).
17. S. F. Brockington, R. H. Walker, B. J. Glover, P. S. Soltis, D. E. Soltis, Complex pigment evolution in the Caryophyllales. New Phytol. 190, 854–864 (2011).
18. Y. C. Chang, Y. C. Chiu, N. W. Tsao, Y. L. Chou, C. M. Tan, Y. H. Chiang, P. C. Liao, Y. C. Lee, L. C. Hsieh, S. Y. Wang, J. Y. Yang, Elucidation of the core betalain biosynthesis pathway in Amaranthus tricolor. Sci. Rep. 11, 1–13 (2021).
19. B. Pucker, N. Walker-Hale, W. C. Yim, J. Cushman, A. Crum, Y. Yang, S. Brockington, Evolutionary blocks to anthocyanin accumulation and the loss of an anthocyanin carrier protein in betalain-pigmented Caryophyllales. bioRxiv, 2022.10.19.512958 (2022).
20. G. Polturak, A. Aharoni, “La Vie en Rose”: Biosynthesis, Sources, and Applications of Betalain Pigments. Mol. Plant 11, 7–22 (2018).
21. N. Walker-Hale, M. A. Guerrero-Rubio, S. F. Brockington, Multiple transitions to high l-DOPA 4,5-dioxygenase activity reveal molecular pathways to convergent betalain pigmentation in Caryophyllales. New Phytol. 247, 341–357 (2025).
22. S. Lopez-Nieves, Y. Yang, A. Timoneda, M. Wang, T. Feng, S. A. Smith, S. F. Brockington, H. A. Maeda, Relaxation of tyrosine pathway regulation underlies the evolution of betalain pigmentation in Caryophyllales. New Phytol. 217, 896–908 (2018).
23. A. Timoneda, T. Feng, H. Sheehan, N. Walker-Hale, B. Pucker, S. Lopez-Nieves, R. Guo, S. Brockington, The evolution of betalain biosynthesis in Caryophyllales. New Phytol. 224, 71–85 (2019).
24. J. W. Clark, Genome evolution in plants and the origins of innovation. New Phytol. 240, 2204–2209 (2023).
25. P. S. Soltis, D. B. Marchant, Y. Van de Peer, D. E. Soltis, Polyploidy and genome evolution in plants. Curr. Opin. Genet. Dev. 35, 119–125 (2015).
26. D. L. Des Marais, To betalains and back again: A tale of two pigments. New Phytol. 207, 939–941 (2015).
27. C. A. Schenck, L. Busta, Using interdisciplinary, phylogeny-guided approaches to understand the evolution of plant metabolism. Plant Mol. Biol. 109, 355–367 (2022).
28. J. F. Walker, Y. Yang, T. Feng, A. Timoneda, J. Mikenas, V. Hutchison, C. Edwards, N. Wang, S. Ahluwalia, J. Olivieri, N. Walker-Hale, L. C. Majure, R. Puente, G. Kadereit, M. Lauterbach, U. Eggli, H. Flores-Olvera, H. Ochoterena, S. F. Brockington, M. J. Moore, S. A. Smith, From cacti to carnivores: Improved phylotranscriptomic sampling and hierarchical homology inference provide further insight into the evolution of Caryophyllales. Am. J. Bot. 105, 446–462 (2018).
29. K. Sugahara, K. Kitao, T. Watanabe, T. Yamagaki, Imaging Mass Spectrometry Analysis of Flavonoids in Blue Viola Petals and Their Enclosure Effects on Violanin during Color Expression. Anal. Chem. 91, 896–902 (2019).
30. M. Farzad, R. Griesbach, M. R. Weiss, Floral color change in Viola cornuta L. (Violaceae): a model system to study regulation of anthocyanin production. Plant Sci. 162, 225–231 (2002).
31. W. Verweij, C. Spelt, G.-P. Di Sansebastiano, J. Vermeer, L. Reale, F. Ferranti, R. Koes, F. Quattrocchio, An H+ P-ATPase on the tonoplast determines vacuolar pH and flower colour. Nat. Cell Biol. 10, 1456–1462 (2008).
32. K. Yoshida, M. Mori, T. Kondo, Blue flower color development by anthocyanins : from chemical structure to cell physiology. Nat. Prod. Rep. 26, 884–915 (2009).


