Gather and Sow: March 2026

We observe two holidays in this March 2026 issue of Gather and Sow: St. David’s Day (March 1st) and Women’s History Month (all of March), which coincides with International Women’s Day on March 8th.
Leeks are a traditional symbol of St. David’s Day, and their morphology is the topic of this issue’s Food for Thought: Ay, leeks is good!
Women’s history month is a time to recognize the contributions of women to American history, society, and culture. Too often women’s accomplishments have gone unsung. The same is true for the “female” parts of flowers. We rectify this by celebrating extraordinary flower parts in corn and the saffron crocus. All celebrations deserve cake, and we offer a recipe for one that combines corn and saffron in this month’s Botany Lab of the Month: Honoring female (plant) anatomy with saffron polenta cake.
We conclude with research and media highlights in Baker’s Dozen and Gleanings. Alongside several recent research articles, we have included a few classic papers by two matriarchs of plant biology, Katherine Esau and Barbara McClintock, in honor of Women’s History Month.
Food for Thought
Ay, leeks is good
LUELLEN …and I do believe your majesty takes no scorn to wear the leek upon Saint Tavy’s day.KING HENRY V I wear it for a memorable honour; For I am Welsh, you know, good countryman.Shakespeare, Henry V, act 4, scene 7
Break out the daffodils and leeks! March 1 is St. David’s Day, the feast day of the patron saint of Wales when many wear daffodils in their lapels and leeks in their caps to celebrate their national heritage. While daffodils bring a bright pop of cheer to early spring, they are not at all edible. Leeks, though, are delicious, especially in stews and roasts made to banish the late winter chill of a fickle March. And if you have a diamond-encrusted leek brooch, as Princess Catherine of Wales has, you can wear the leek without the stink and save the real leeks for the kitchen.

Leeks, along with onions and garlic, are in the genus Allium in the family Amaryllidaceae. (Daffodils are in the same family). Like many other monocots, they have long slender leaves striped with parallel veins. Also common in monocots, and sometimes confusing, is the way their leaves develop. The white part of a leek looks like a big solid cylinder composed of concentric leaf tubes — and essentially it is. Each new leaf develops at the center of the cylinder and completely surrounds the next little mound of tissue that will develop into a leaf.

This process is easy to see if you slice the root end of the leek in half lengthwise, as close to the center as possible, and examine the innermost leaves. These leaves will be short, lengthening as they develop. In mature leaves, the top part of the tube opens up into a flat green blade. The pattern of leaves enclosing leaves at the base is evident in many monocot houseplants, such as peace lilies and orchids. It is also easy to see in corn plants as well as onions.

One of the most fun aspects of leek morphology, however, is their contractile roots. Look closely at the roots, and you will see that many are ribbed horizontally like a pushed-up sleeve or an accordion. Such ribbed roots have already done their job, contracting to pull the plant down deeper into the soil. No wonder those resourceful leeks were chosen as a national symbol. As Fluellen says later on in the play, “Ay, leeks is good.”
Botany Lab of the Month
Honoring female (plant) anatomy with saffron polenta cake
Corn plants hold the botanical record for longest styles. We haven’t seen this officially published anywhere, but we did ask around, and the surveyed plant experts couldn’t think of another species that can beat it. Corn styles are usually called “silks”, the threads that erupt from the tips of corn ears, and they can be up to a foot long. There are, of course, species with more massive flowers, but in all of them the style–the structure through which a pollen tube must grow to reach an ovule–is comparatively short.
In general humanity is pretty good at celebrating the extreme sizes of natural things—the biggest or smallest plant, leaf, flower, or seed , for example. For some reason, however, the flower parts that we call “female” (for convenience), including the style, have escaped allometric scrutiny despite their remarkable and important function. It feels depressingly unsurprising that we take for granted the parts of plants that perform extraordinary reproductive feats and thereby make possible the food we get from grains, seeds, and fruit.
Given the ubiquity of corn (also called maize; Zea mays, Poaceae), the millions of acres planted with it around the world, and its importance to the human diet, we feel like the exceptional biology of the production of each kernel should be more common knowledge.
Similarly, the spice saffron is literally made of dried female flower parts–the stigma, in this case–but we rarely pause to consider that. Saffron “threads” are prized for their unique aroma and gorgeous color, and their anatomical origin should also be appreciated.
Yes, part of a plant’s female anatomy is literally called a stigma. The term’s use in botany dates back centuries and is somehow related to the Latin meaning of the term, referring to a mark, tattoo, or stain. The history is murky (1, 2).
Strictly speaking, it is inaccurate to label plants or their reproductive organs as “male” or “female”. The relevant mechanics in plants–the whole alternation of generations thing, among other genomic complications–cannot be defined by binary sexual terms and their derivatives, like “hermaphrodite”. It is nevertheless useful to borrow gendered language, as metaphors, when we talk about the function of plant reproductive structures (3). We’ll get into this a bit more below.
Here we celebrate the reproductive biology of corn and saffron plants with knowledge and cake (the recipe for saffron polenta cake is below).
Building flowers
A corn silk is technically a fusion of style and stigma, the parts of a flower’s reproductive apparatus that intercept pollen and facilitate fertilization. A close examination of a corn silk reveals numerous small, sticky hair-like extensions (called trichomes) along its length, to which airborne pollen adheres.



Left: Mature ear of corn with decayed silks extending the typical 2-3 inches from the husk. Most of the silk’s length is hidden underneath the husk (photo from (4)). Center: Pollen sticking to sticky trichomes on red silks (photos from (5)). Right: Silks hanging from an immature ear of corn with husks removed. Few of the silks have detached from their respective kernels, indicating that fertilization was largely unsuccessful (photo from (5)).
In many species the stigma and style are more clearly differentiated, hewing more closely to the generic heuristic diagram of flower parts that we all learn first in school. A flower’s stigma and style connect to its ovary. Collectively, these three structures comprise the pistil, also called the gynoecium. The latter term, which translates to “female house”, is a clue that these structures are often referred to as a flower’s female parts.
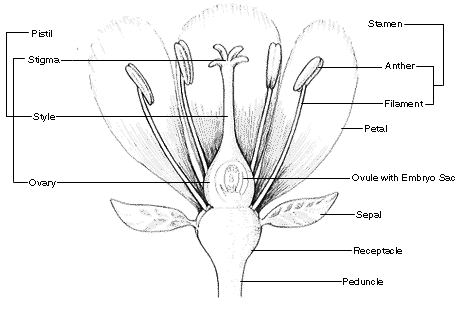
As we mentioned above, plants do not have a sex, but the gendered language is useful to communicate about the function of plant reproductive structures, which is all about transferring genes to the next generation through eggs and sperm. In flowering plants (angiosperms), male function is performed by pollen and the structures that make and deploy it (a flower’s anthers, borne on stamens). Female function is performed by the structures that intercept pollen (the flower’s stigma and style) and house egg cells (the flower’s ovary and the ovules within). After fertilization, female function of the ovary extends to nurturing seeds to maturity and ensuring seed dispersal.
In corn plants, performing female function is a challenging task. Although it takes place an estimated 7 quadrillion times a year, it requires those record-breaking flower parts to accomplish (7). By contrast, saffron crocus flower parts contribute absolutely nothing to female function. The domesticated plants are sexually sterile, and their stigmas are valuable only in our human realm.
Corn silks and the making of a kernel
One of the most dramatic feats in cellular biology takes place inside corn silks: the extremely rapid growth of corn pollen tubes through the entire length of the corn silks (8). A pollen grain will germinate and penetrate the silk surface within minutes of landing on a silk. It then uses nutrients from the silk itself to grow the pollen tube toward the ovule at the astonishing rate of up to 1 centimeter per hour, transferring genetic material (via sperm cells that travel down the pollen tube) and completing fertilization within 24 hours (9).
Corn plants differ from most other grass species (family Poaceae) in that they are monoecious (“house of one”), meaning that they bear distinct “male”and “female” flowers. Since flowers do not themselves have a sex, these differentiated blossoms are properly called “staminate” (having stamens) and “pistillate” (having a pistil) flowers, respectively. Staminate corn flowers are arrayed along the tassels on the top of each corn plant, where they can most effectively shed their pollen into the wind.


Left: Corn plants in a field, each with one or two developing ears of corn near the middle of the plant, and tassels at the top. Image from (10). Right: Tassels on top of a corn plant showing the anthers of staminate flowers. Image from (11).
Each pistillate flower bears a single corn silk. The pistillate flowers are tightly clustered on short, stout stems–the corn cobs–in the developing ears, buried within the husk–the collection of modified leaves that surround each cob and that will someday surround tamales. Each flower’s silk has to extend along the entire length of the cob within the husk and for several inches beyond in order to access wind-borne pollen. The silks of flowers at the bottom of the cob, then, must be longer than those of flowers at the top. If pollination is successful, each pistillate flower will develop into a single kernel of corn. Each kernel is an individual fruit, tightly bonded to the seed within. After fertilization, the silk detaches from the ovary (kernel), but it remains trapped within the husk.
Why must silks be so long? Much of the length is simply attributable to the size and structure of the corn ear. That large ear size is a product of a few millennia of dedicated plant domestication and selective breeding started by early Mesoamerican farmers (12). Part of the length, too, must be because corn plants are so lush and bear their ears relatively low on the plant, shielded from falling pollen by the long, broad leaves above them on the stem. Many other grass species put their pistil-bearing flowers on a tall stalk above the leaves, so the pollen in the wind hits the flowers before the leaves. Imagine a field of wheat or oats with heavy inflorescences swaying in the breeze. Not so with corn. The silks may have to compensate for their position with surface area.
The profusion of corn silks can make for tedious shucking of fresh corn on the cob, picked when the kernels are immature and succulent and still retain vestigial flower parts. Next time you shuck an ear of fresh sweet corn, pay attention to those impressive styles. Removing silks can be an annoying aspect of corn cob preparation, but perhaps the knowledge that they are extraordinary will help make the job less odious. Just think of it as breaking down social stigmas.
Saffron
Saffron crocus (Crocus sativus, Iridaceae) flowers look more like the textbook diagram than corn flowers do, but they have a few quirks too.
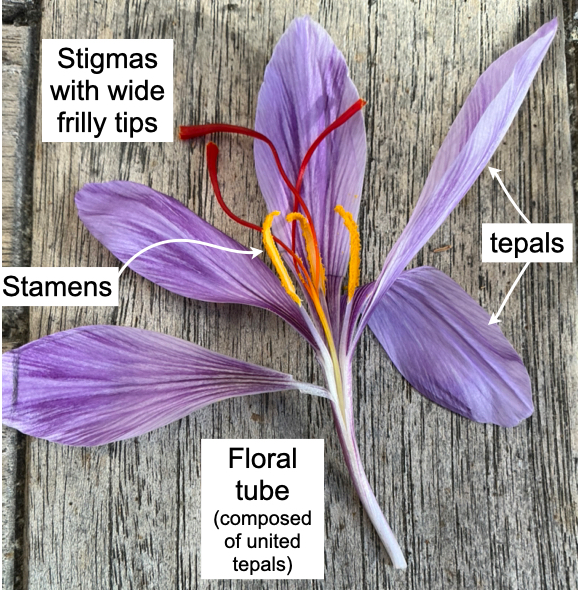

Left: The aboveground portions of a saffron crocus flower, labeled, from a flower grown and photographed by K. A. Preston. Right: Saffron crocus illustration (from Wikipedia) showing the belowground position of the ovary, above the corm. The flower’s style travels from the stigmas, through the floral tube, and belowground to reach the ovary.
All of the nearly one hundred species in the genus Crocus have three orange stigmas, usually with wide, frilly tips, but the pendulous length, deep color, and characteristic aroma of saffron is only found in C. sativus and its wild progenitor, C. cartwrightianus (13, 14). The large, aromatic stigmas on saffron’s ancestor were likely adaptations to attract insect pollinators, mainly bees (15, 16).



L to R: Saffron crocus (Crocus sativus); Cartwright’s crocus (C. cartwrightianus, image from Wikipedia), the ancestor of saffron; and snow crocus (C. sieberi, photo from Dave’s Garden), a popular choice for gardeners. Crocus species each have three yellow stamens and three orange stigmas.
Each crocus style is also rather long–not corn silk long, yet still impressive–but most of it is hidden. Each broad orange stigma tapers into the top of the style. The rest of the style is occluded aboveground by the tubular base of the tepals (the showy floral parts that are not obviously distinct petals and sepals), and belowground by the sheath of papery modified “tunicate” leaves that protect the ovary. Most species, of course, do not hide their flower ovaries underground, but this works for Crocus. If pollination is successful and a fruit begins to form, the short scape (flower stalk, or peduncle) that connects a Crocus flower to its corm pushes the developing fruit above ground.

To harvest saffron to make the spice, each stigma must be hand-plucked from its freshly-opened flower and quickly dried. The high cost of dried saffron is explained by this laboriousness of harvest, combined with the land acreage that must be dedicated to the perennial crocus plants that bloom but once a year. The harvest of crocus stigmas for color and flavor in the Mediterranean region dates at least to the Bronze Age (17–19). The process of bringing saffron under cultivation has involved very little modification from the wild variants. The ability of farmers to further push stigma evolution through domestication is limited by the unique genetics of the saffron crocus. In what must be one of the greatest ironies among edible plants, this celebrated spice, which is made of a charismatic female flower apparatus, comes from a species that cannot sexually reproduce (20).

As with many food plants, the saffron crocus is a happy genetic accident. It was created when a hapless ancient Greek C. cartwrightianus left an extra copy of half of its genome in its seeds, instantly resulting in the brand new triploid species C. sativus (20). The stigmas of C. sativus are larger and more aromatic than those of its ancestor, which would have made the C. sativus variants stand out from their C. cartwrightianus brethren at harvest time (20). The extra set of genes improved C. sativus as a spice but wreaked havoc with its reproductive biology. C. sativus cannot produce viable seeds. The inability to sexually reproduce limits new genetic changes–new agronomic possibilities–to random mutations that happen when the plant vegetatively (asexually) replicates itself. Like most perennial underground storage structures, crocus corms spread underground by creating clones of themselves, called cormels. Cormels are separated from the mother corm and are the only way to propagate saffron crocuses.
Saffron Polenta Cake
The silks on an ear of corn will be dried and withered by the time the kernels mature into the hard, dry fruits that can be popped for popcorn or ground for cornmeal, but it is worth remembering the extraordinary flowers they once were. We can appreciate the juxtaposition of something that we dismiss as pedestrian—cornmeal—with the heroism of the structures that produced it. Similarly, pairing cornmeal, in polenta form, with saffron is a study in contrasts and similarities. The common pantry staple is combined with one of the most valuable spices on Earth. Nonetheless, they share a history of exceptional female flower function. And they are both monocots! But that is a story for another day.
Our saffron polenta cake recipe is inspired by David Lebovitz’s recipe for polenta cake with olive oil and rosemary from his book Ready for Dessert.
Ingredients
- 1 tablespoon plus 1/2 cup unsalted butter, at room temperature
- 2 tablespoons plus 1 cup polenta or stone-ground corn meal
- 1 cup all-purpose flour
- ½ teaspoon crushed saffron threads
- 2 teaspoons baking powder
- 1 teaspoon salt
- 1/2 cup olive oil
- 6 large eggs, at room temperature
- 1 1/3 cups sugar
Instructions
- Gently heat olive oil and crushed saffron in a stainless steel bowl set over a pan of simmering water, until the olive oil is warm and the saffron has infused into it, about 5 minutes. Set the bowl with the infused olive oil aside to cool.
- Preheat the oven to 350°F. Grease a 10-inch bundt or tube pan with 1 tablespoon of butter, then dust with the 2 tablespoons polenta or corn meal.
- In a small bowl sift together the flour, 1 cup of polenta, salt, and baking powder.
- In a stand mixer fitted with a paddle, beat the ½ cup butter and sugar until light and fluffy, 3 to 5 minutes. Drizzle in the saffron-infused olive oil. Add in the eggs one at a time, mixing before each addition. Stir in the flour mixture until just incorporated.
- Scrape the batter into the prepared cake pan and smooth the top. Bake for 40 minutes or until a toothpick inserted into the center comes out clean. Let the cake cool for 30 minutes before inverting it onto a serving plate.
Baker's Dozen and Gleanings
Baker’s Dozen
To our usual roundup of recent papers that caught our attention, we have added four very old ones in honor of Women’s History Month. We celebrate significant contributions to botany made by a couple of women who, nevertheless, persisted.
- Yan, Y., Tian, B., Li, X., Wang, Y., Shi, Y., Jiang, K., ... & Li, P. (2026) The polyphenol trade‐off: leaf protection vs fruit attraction in apples driven by the phlorizin glycosyltransferase PGGT1. New Phytologist. Apple trees express polyphenols in their leaves to reduce herbivory. The primary polyphenol is also expressed in their fruits, but is converted to a version that does not deter seed dispersers. Yan et al. uncovered the genetic basis for this conversion.
- Shen et al. (2026) OsWRI1a coordinates systemic growth responses to nitrogen availability in rice. Science 391: 937-945. Rice plants are the subject of this work that provides new insight into the genetic mechanisms responsible for coordination of above- and belowground responses to nitrogen availability. Commentary is here.
- Jørgensen et al. (2026). Postdomestication selection of MKK3 shaped seed dormancy and end-use traits in barley. Science 391: 90-95. How does a barley seed know when it’s time to sprout (break dormancy)? This work helps us understand that.
- Guo, Y., Jayakodi, M., Himmelbach, A., Ben-Yosef, E., Davidovich, U., David, M., ... & Mascher, M. (2025). A haplotype-based evolutionary history of barley domestication. Nature, 647(8090), 680-688. Speaking of barley, we missed this one back in September. Domesticated barley is a genetic mosaic of wild and cultivated populations. Like barley in your St. Patrick’s Day stew, this paper adds some bits to chew on.
- Hu, Y., Ma, Q., Ye, X., Xiao, Y., Song, X., Wu, J., & Zhao, Z. (2026). Reduced resource competition from the stem when brassinosteroid biosynthesis is inhibited enhances curd yield in cauliflower. Journal of Experimental Botany, 77(4), 1107-1122. The authors find that cauliflower stems use resources that could be allocated to the curd, the part we eat. Spraying stems with two growth inhibitors enhanced curd production.
- Gautam, B., Jarvis, B. A., Esfahanian, M., McGinn, M., Williams, D., Liu, S., ... & Sedbrook, J. C. (2026). Creating a new oilseed crop, pennycress, by combining key domestication traits using CRISPR genome editing. Nature Plants, 12(1), 74-87. A stinky cover crop with unsuitable oil in its seeds can be edited to produce oil similar to canola. These plants can do double duty as cover and edible crop.
- Garsmeur, O., Rio, S., Pompidor, N., Lipzen, A., Hervouet, C., Durand, T., ... & D’hont, A. (2025). The genomic footprints of wild Saccharum species trace domestication, diversification, and modern breeding of sugarcane. Cell, 188(25), 7252-7266. The domestication history of sugar cane has been hard to work out because it probably involved hybridization with several wild species in New Guinea and nearby regions. This study uses variation in transposable elements (“jumping genes”) to posit an additional ancestor that has not yet been identified in the wild.
- Sánchez-del Pino et al. (2026). The current state of our knowledge of the domestication and evolution of the grain amaranths: a critical standpoint. Annals of Botany 137: 609-630. Independent domestications in Asia and the Americas, involving multiple species.
- Dominguez Mendez and Studer (2026). Is more better? Polyploidy in crops with diverse end uses and the potential for future applications. Annals of Botany 137: 631-639. A cogent review of the impacts of polyploidy in crops and the potential afforded by all that extra genetic material
- Burton et al. (2026). Phylogenetics and evolution of Digitaria grasses, including cereal crops fonio, raishan and Polish millet. Annals of Botany 137: 141-151. Why aren’t these in my pantry?
- Wakib et al. (2026). Intraspecific variation and phenotypic plasticity of olive varieties in response to contrasting environmental conditions. AoB PLANTS 17: plaf060. Insight into how olive trees make a living across the Mediterranean and how varieties may respond to climate change.
- Anyone who has ever studied plant anatomy has benefited from the painstakingly meticulous work of Katherine Esau (1898-1997). Yes, she was 99 when she died and kept publishing until she was 93. She was an overachiever in every respect. Fleeing what is now Ukraine and ending up in California is just the beginning. When people couldn’t handle her 700-page masterpiece (Plant Anatomy, 1953) she created a shorter version, The Anatomy of Seed Plants (1960), to be used as a textbook. In addition to her indispensable texts, we have two favorite papers:
- Esau, K. (1936). Ontogeny and structure of collenchyma and of vascular tissues in celery petioles. Hilgardia, 10(11), 429-476. Esau pulled the strings out of celery petioles (“stalks”) and stretched them until they broke. She found that the vascular tissue is much weaker than the strands of collenchyma, which are composed of living cells with thick walls.
- Esau, K. (1940). Developmental anatomy of the fleshy storage organ of Daucus carota. Hilgardia, 1940, Vol. 13, 175-209 ref. bibl. 38. Esau breaks down all the cool parts of a carrot root, including the parts you can see while eating them. Sadly, this paper is nearly impossible to find without going to a university library in person.
- Undoubtedly you have heard the story of Barbara McClintock (1902-1992), famous for discovering “jumping genes” in corn (and not being believed). But did you know that she and a graduate student (Harriet Creighton) were the first to confirm that during meiosis (gamete formation), chromosomes physically exchange segments (often called “crossing over”)? Fruit fly geneticists had been able to infer this process, but not watch it happen. By using a variety of corn with a conspicuous knob on one end, McClintock and Creighton were able to verify the physical exchange of chromosome segments.
- Creighton, H. B., & McClintock, B. (1931). A correlation of cytological and genetical crossing-over in Zea mays. Proceedings of the National Academy of Sciences, 17(8), 492-497.
- Creighton, H. B., & McClintock, B. (1935). The correlation of cytological and genetical crossing-over in Zea mays. A corroboration. Proceedings of the National Academy of Sciences, 21(3), 148-150. Four years later, they had to publish more data because, guess what? People doubted them. The introduction to the new paper is understandably impatient. Originally, “...the authors felt it unnecessary to add to the ever-increasing amount of published work merely to record more evidence of the same nature without supplying anything essentially new or advancing…. However, we now feel forced to add more data merely to counteract any suspicion that the evidence previously presented constituted insufficient proof.”
Gleanings
- The Throughline podcast had an episode on the history of chocolate back in December.
- A recent episode of the In Defense of Plants podcast interviewed the founders of the Let’s Botanize! plant educational endeavour.
- Beronda Montgomery’s book When Trees Testify received a positive book review in Science and so far Katherine has been enjoying her own copy very much.
- Can Taylor Swift help people notice plants? Maybe?
References
1. Opening Up on the Names of Flower Parts. https://www.merriam-webster.com/wordplay/origin-of-flower-part-names.
2. A Grammatical Dictionary of Botanical Latin. https://www.mobot.org/mobot/latindict/keyDetail.aspx?keyWord=stigma.
3. J. R. Pannell, Sex, sexes, sex roles, and gender in land plants. Am. J. Bot. 110, e16195 (2023).
4. Nielsen, Robert L., Unusually Long Silks in Corn. https://www.agry.purdue.edu/ext/corn/news/timeless/LongSilks.html.
5. Nielsen, Robert L., A Fast & Accurate Pregnancy Test for Corn (Purdue University). https://www.agry.purdue.edu/ext/corn/news/timeless/EarShake.html.
6. Parts of a Flower: An Illustrated Guide | AMNH, American Museum of Natural History. https://www.amnh.org/learn-teach/curriculum-collections/biodiversity-counts/plant-identification/plant-morphology/parts-of-a-flower.
7. C. McNinch, K. Chen, T. Dennison, M. Lopez, M. D. Yandeau-Nelson, N. Lauter, A multigenotype maize silk expression atlas reveals how exposure-related stresses are mitigated following emergence from husk leaves. Plant Genome 13, e20040 (2020).
8. A. Lausser, I. Kliwer, K. Srilunchang, T. Dresselhaus, Sporophytic control of pollen tube growth and guidance in maize. J. Exp. Bot. 61, 673–682 (2010).
9. A. Krichevsky, S. V. Kozlovsky, G.-W. Tian, M.-H. Chen, A. Zaltsman, V. Citovsky, How pollen tubes grow. Dev. Biol. 303, 405–420 (2007).
10. D. Murray, Record corn exports and production lowered in latest WASDE, High Plains Journal (2025). https://hpj.com/2025/07/23/record-corn-exports-and-production-lowered-in-latest-wasde/.
11. Nielsen, Robert L., Tassel Emergence and Pollen Shed. https://www.agry.purdue.edu/ext/corn/news/timeless/Tassels.html.
12. M. C. Stitzer, J. Ross-Ibarra, Maize domestication and gene interaction. New Phytol. 220, 395–408 (2018).
13. S.-S. Kazemi-Shahandashti, A. El-nagish, D. Harpke, T. Heitkam, Ancient Artworks and Crocus Genetics Both Support Saffron’s Origin in Early Greece. Front. Plant Sci. 13 (2022).
14. Z. Nemati, D. Harpke, A. Gemicioglu, H. Kerndorff, F. R. Blattner, Saffron (Crocus sativus) is an autotriploid that evolved in Attica (Greece) from wild Crocus cartwrightianus. Mol. Phylogenet. Evol. 136, 14–20 (2019).
15. K. Lunau, S. Konzmann, J. Bossems, D. Harpke, A Matter of Contrast: Yellow Flower Colour Constrains Style Length in Crocus species. PLOS ONE 11, e0154728 (2016).
16. T. Pastor-Férriz, M. De-los-Mozos-Pascual, B. Renau-Morata, S. G. Nebauer, E. Sanchis, M. Busconi, J.-A. Fernández, R. Kamenetsky, R. V. Molina, Ongoing Evolution in the Genus Crocus: Diversity of Flowering Strategies on the Way to Hysteranthy. Plants 10, 477 (2021).
17. S. S. Kazemi-Shahandashti, L. Mann, A. El-nagish, D. Harpke, Z. Nemati, B. Usadel, T. Heitkam, Ancient Artworks and Crocus Genetics Both Support Saffron’s Origin in Early Greece. Front. Plant Sci. 13 (2022).
18. M. Negbi, “Saffron cultivation: past, present, and future prospects” in Saffron (CRC Press, 1999).
19. R. Dewan, Bronze Age Flower Power: The Minoan Use and Social Significance of Saffron and Crocus Flowers. 5 (2015).
20. T. Schmidt, T. Heitkam, S. Liedtke, V. Schubert, G. Menzel, Adding color to a century-old enigma: multi-color chromosome identification unravels the autotriploid nature of saffron (Crocus sativus) as a hybrid of wild Crocus cartwrightianus cytotypes. New Phytol. 222, 1965–1980 (2019).

