Gather and Sow: May 2026

Coconut palms grow some of the biggest seeds on the planet, and vanilla seeds, from an orchid, are among the smallest. They are both delicious! They are also the subjects of this month’s newsletter. Just in time for Mother’s Day (May 10th this year), in this month’s Food for Thought essay, we make the case that Mother’s Day celebrations should include more coconut. The large seed size of coconuts, and the small size of orchid seeds, are indicative of their respective parenting philosophy. In this month’s Botany Lab of the Month we describe coconut and vanilla fruit and seed morphology in the course of making cookies (Extreme Monocot Cookies). The newsletter concludes with Gleanings and Baker’s Dozen, our roundup of relevant recent media and journal articles.
Food for Thought
Coconuts for Mother's Day
There is a good chance that any given Mother’s Day celebration will involve orchids. Orchid blossoms, as cut flowers or houseplants, are among the more popular choices of Mother’s Day gifts. And if vanilla is one of the ingredients in a sweet treat at a Mother’s Day brunch or dessert, orchids are involved again, in the form of vanilla “beans” and the flavoring derived from them. Orchid flowers and vanilla are lovely, so it makes sense to include them on Mother’s Day for aesthetic reasons. From a purely biological perspective, though, orchids are, at best, ironic choices as maternal symbols. Paragons of nurturing they are not. If we had to choose one plant to acknowledge extraordinary maternal investment in each offspring, Mother’s Day would be coconut-themed.

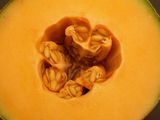
The rough measure of a plant’s maternal investment in an offspring is seed size, and this varies dramatically across the global flora, spanning 11.5 orders of magnitude (1). Coconuts are among the heaviest seeds on the planet, and all of the smallest are from orchids. A person strolling on a tropical beach where coconut palms grow needs to be careful of falling coconuts, each a genuine mortal hazard. In contrast, a person could be pelted with thousands of orchid seeds and not even notice. The tiny black specks in a respectable vanilla ice cream or custard are seeds from the vanilla orchid (Vanilla planifolia), and those are actually quite large for an orchid.

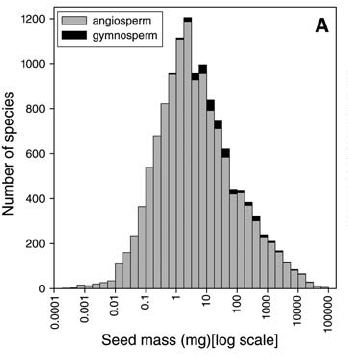
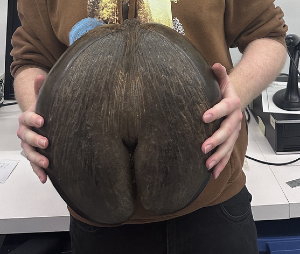
Left: small orchid seeds (not vanilla) next to a British 20p (from Kew’s blog); center: frequency distribution of seed mass for 12,987 species (Fig. 1A from (1)); right: the double coconut (Lodoicea maldivica (2)), the largest seed in the world, sadly inedible (image from Wikipedia)
So why are orchid seeds so small, and coconut seeds so big? The short answer is because the “mother” plant said so (Strictly speaking, by “mother” we mean the ovule-bearing plant that nurtures seeds to maturity. As we discussed in the March newsletter, plants don’t have sexes, but it is convenient to use gendered language to describe sexual functions in plants (5)). The size of a typical seed is under maternal control (6–10). In most species a mother plant produces more than one seed, and she must hedge her bets–the gamble that the embryo within any given seed will reach adulthood and produce seeds of its own–across all her offspring. Producing seeds is energetically and nutritionally expensive for the mother plant, and she only has a certain amount of energy and nutrients to allocate to seed production. She can therefore either invest a large fraction of that allocated amount into each of a few large seeds, or she can invest a smaller fraction into each of several smaller seeds. There is cool math that describes this seed size vs. number tradeoff with reasonably well supported predictions about how the mother plant should allocate her reproductive effort across her seeds under different scenarios (7, 11).
Across all species, however, there are a few trends in the relationship between seed size and a species’ life history and ecology–its characteristics, the place where it lives, and the other organisms with which it interacts (1, 6, 12). A seed has a few jobs: (A) to disperse, hopefully to a place suitable for the embryo within it to grow; (B) to protect the embryo until conditions are right for germination; and (C) to aid the embryo’s transition to seedling upon germination (10). Each of these job components can push seed size toward being large or small. Seeds that are wind-dispersed, for example, must be small and light (13–17). Some even have wing-like appendages on their seed coats to catch the wind. Seeds that are dispersed by animals or other means vary in size, depending on each seed’s complement of protective structures or nutritive reserves. The seeds of most species store a certain amount of nutritive tissue (endosperm or cotyledon, depending on the taxon) to feed the seedling after germination until it can photosynthesize on its own. The amount of provisioning is related to the environment in which the seed will germinate and the characteristics of the plant it will become.

Bigger plants tend to produce bigger seeds. This trend seems to be especially true among closely-related species. Think about the seeds of familiar fruit species in the rose family (Rosaceae): strawberry plants are herbaceous and have very small seeds. Roses, service berries, raspberries and blackberries grow on woody shrubs or vines, and their seeds are a bit bigger than those of strawberries. The biggest seeds are in the tree species within the family: almonds, peaches, plums, nectarines, apricots, cherries, apples, pears, loquats, quince, medlar. The palms (family Arecaceae) are the only tree-sized woody monocots, so large plant size may be one factor pushing large seed size in coconuts.
Seeds tend to be larger for species that germinate under stressful conditions: in shade, or in saline or nutrient-poor soils, or in dry places (6, 7, 11, 12, 18–21). Several plant families, for example, have species that became mangrove trees, living in coastal areas in wet soils of various salinities. In every case, seed size is bigger in the mangrove species than in its closest relatives (1). Under stressful conditions, a seedling may require more time or support until it can produce enough of its own food, so the seed supplies a large amount of nutritive reserve, which increases seed size. Coconuts, of course, inhabit sandy tropical beaches. This sounds pretty appealing to us, but only because we’re not seedlings, so it’s a good bet that environmental stress has helped push seed size in coconuts.
Most orchids and some other plant taxa have arrived at an alternative arrangement for fueling their seedlings: taking advantage of fungus (22–27). Instead of relying on nutrients packed into a seed, the embryo becomes a parasite on a fungus, which in turn gets its nutrients and energy from decaying organic matter or by being a parasite itself upon other plants (28). After seedling establishment these mycotrophic (fungus parasitizing) plants may become independent of their fungal symbiont, or they may maintain the relationship, often repaying the fungus with sugar from photosynthesis. The set of fungi that help an orchid germinate may be different from those that form symbiotic relationships with it as an established plant (29). Many plants form symbiotic relationships with soil fungus (mycorrhizae), in which the fungus receives photosynthate in exchange for soil nutrients, but only a few plants forego seed nutrient reserves and rely on fungal support for seedling establishment. Without the burden of nutritive tissue, seeds can get very small, and a mother plant can make millions of them.
Most orchid seeds are dry and are wind-dispersed, but vanilla seeds are relatively big for an orchid and are coated in a sticky resin, the origin of the delicious vanilla aroma. The vanilla fruit (“bean” or “pod”) splits when ripe, exposing the resinous seeds to the air. The vanilla aroma likely serves as an attractant for the seed-dispersing animals (30–35), who eat the seeds and resin. Passing through an animal’s gut seems to enhance seed germination of vanilla seeds (36). If the seeds are not directly consumed, the resin might act as a glue to adhere seeds to the bodies of visiting animals, who may carry the seeds to other parts of the forest canopy. Like many orchids, Vanilla planifolia plants are epiphytic, meaning they sprout and grow on the trunks and branches of other woody plants in the forest (34).

Coconuts, on the other hand, are dispersed by floating across the ocean. Their large size and a morphology designed for buoyancy (hollow seed cavity, protective husk) helps them make the voyage (37–40). If a coconut doesn’t get picked up by the tides, then it stays where it falls, close to home, which is apparently exactly where mother wants it.
Botany Lab of the Month

The orchid family (Orchidaceae) and palm family (Arecaceae)--home of vanilla and coconuts, respectively–are both in the monocots, a major clade that accounts for about a sixth of flowering plants. It is interesting that the entire range of seed sizes in extant land plants can be found in that one clade, and it is delightful that representatives from the large and small tail ends of that seed size distribution are food plant species. Furthermore, in our opinion, vanilla and coconut taste good together. We combine them here in a cookie recipe. Every single one of the ingredients in this recipe is derived from a monocot except the salt and baking powder (which is usually bulked up with corn starch, also a monocot product). We provide instructions for using fresh whole ingredients that highlight typical monocot features, but if you are short on time or ingredients, cupboard substitutes will work just as well. These cookies are vegan and gluten-free. Because they are low in sugar and high in fruit, they bake up soft rather than crisp or chewy.
Ingredients
½ lb ripe bananas (about 2 medium sized fruits)
2 T coconut oil, softened
2-in long segment of fresh ginger, sliced (or ½ teaspoon dried ginger)
¼ teaspoon salt
1-in long piece of vanilla “bean” (or ½ teaspoon vanilla extract)
2T of a monocot-derived sugar such as cane, date, or coconut sugar
1 ½ cups shredded coconut, extracted from the shell of a mature (not green) fresh coconut (or 1 cup dried unsweetened shredded coconut), divided
1 ½ cups rolled oats, divided
½ teaspoon baking powder
¾ cups dried pineapple, preferably unsweetened, coarsely chopped
Oven 350º F
Makes approximately 2 dozen cookies
Instructions
- Before peeling the bananas, look at the flat rough patch on one end of the fruit (opposite the “handle”, the short stem that connected the banana to its branch) to see the ring of scars left behind by the various parts of the fallen banana flower.



Left: Pendulous yellow banana flowers open under the first set of purple bracts on the large banana inflorescence. Each set of flowers is called a "hand" or "bunch" and is attached to its own branch of the inflorescence. The tubular petals will fall off of the developing fruit. Photo by F. Fleischmann. Center: a hand of bananas in a market in Cambodia. Notice the flat, brown scar opposite the stem on each fruit, were the floral parts were attached. Photo by L. Osnas-Paguio. Right: fruit from a peeled Cavendish banana. Notice the three sections (locules) and undeveloped seeds. Photo by J. Osnas
- Peel the bananas and observe that the flesh falls into three longitudinal segments. Sometimes the undeveloped seeds can be seen as small black dots.
- Mash the bananas into a chunky paste and measure out ¾ cup to use in the recipe.
- If using fresh ginger, take a moment to notice its typical monocot properties. Ginger “root” is an underground stem (rhizome), with branches and encircling leaf scars. Like other monocots, the stem is very fibrous. Slice about 2 inches of the ginger.

- In a food processor, combine the bananas, coconut oil, ginger, salt, and sugar. Process until the ginger is nearly pureed.
- With a paring knife, open up the vanilla bean, which is the dried and fermented fruit of the vanilla orchid. Scrape the miniscule seeds into the banana and oil mixture in the processor. These seeds are the smallest you will ever find in your kitchen and are among the smallest on the planet. They don’t even have any endosperm to support the tiny embryo.


- If you are using a fresh coconut, you may now turn to one of the largest seeds on the planet. A coconut has abundant tasty endosperm. Follow the instructions below to crack the fruit wall of the coconut and reveal the seed. Then grate the flesh and measure 1 ½ cups into a mixing bowl. Alternatively, measure out 1 cup dried unsweetened grated coconut.
- The hard shell of a coconut as typically found in a store is the innermost part of the fruit wall that surrounds the single large seed inside.


A coconut is a drupe, like (non-monocots) avocado, olives, and stone fruits (peach, almond, cherry, and plum). This means that the outermost part of the fruit is either fleshy or fibrous (coconut fruits are fibrous), and the innermost part of the fruit is a very hard shell that protects the single seed within. This is the “stone” or “pit” or “shell”. Some of the fibers (called “coir”) from the outermost part of the fruit are clinging to the shell of this coconut from the grocery store. The eye on the left looks different and soft. That is the one to puncture. In the image on the right, we see that the small, cylindrical embryo came out when we punctured the eye (it’s sitting on top of the coconut).
- Use a metal skewer or table knife to pierce the softest of the three “eyes” on top of the fruit. This is where the coconut seedling would have emerged. Sometimes the short cylindrical embryo will come out along with the eye.
- Drain the coconut water (liquid endosperm that hasn’t solidified into hard, white endosperm yet) from the hole and save it for another use.
- Steady the coconut on a cutting board by nestling it in a kitchen towel and hit it firmly with a mallet or a hammer all along its equator, rotating all the way around. The shell will eventually crack and may then be pried open to reveal the seed.
- The flesh of the coconut seed is its solid cellular endosperm. It is pressed tightly against the inside of the fruit wall but can be pried away in chunks with a table knife. It sometimes helps to break up the shell more. The seed coat will remain on the outside of the chunks and does not need to be removed.

- Add half of the oats and half of the coconut to the banana mixture and process. The mixture will remain a bit chunky.
- Combine the remaining oats and coconut in a bowl and add the baking powder and chopped pineapple. Stir the banana mixture into this dry mixture.
- Line a baking sheet with parchment paper. Use about 2 tablespoons of dough for each cookie. Drop the dough onto the parchment paper and gently flatten the mound. They will not spread, so they can be placed close together. Bake 18-20 minutes.
Gleanings and Baker's Dozen
Gleanings
- The botany class that Darwin took at Cambridge is being offered again.
- Ricardo Kreibel will give a talk entitled “Moving Flowers: Evolution of Floral Form, Function, and Pollination in Salvia” on May 14th for the California Botanical Society’s speaker series. Anybody can view via zoom. Salvia includes culinary herbs sage and rosemary.

- What’s the secret to a great cup of tea? Amber Dance has written a worthwhile article about tea (from Camellia sinensis) in Annual Review’s Knowable magazine.
- This person has a cool job: stewardship of 1200 years of data on cherry blossom flowering dates in Japan.
- Jeremy Cherfas provides insight into how collard greens made their way to the American South in the latest episode of his Eat This Podcast: “Collards: A Moroccan Mystery”.
- Andrielle Swaby brings her chops as an evolutionary biologist to her side gig as an artist and plushie designer under the admirable label Phylogeny Unlimited. She designed the biologically accurate Paleozoic Pals for the Paleontological Research Institute. Her Kickstarter for Cenozoic plushies just closed, but if all goes well, you can soon be the owner of a Megatherium or Glyptodon plushie. These giant ground sloths and proto-armadillos were among the megaherbivores that shaped the evolution of several food plant species (we mentioned these in our February newsletter). There’s a recent Nautilus magazine article about the ecosystem consequences of the loss of large mammals.

Baker’s Dozen
- Song, X., et al. (2026) Dynamic chromatin regulatory programs of sucrose and citric acid metabolism during fruit ripening in Citrus. The Plant Cell 38: koag060. Shedding light on how citrus fruits accumulate sugar and acid during ripening.

- Sun, Y., et al. (2026) Concentration-Dependent Regulation of Ginger Growth and Quality by Abscisic Acid: Insights from Integrated Metabolomic and Transcriptomic Analyses. Plants 15: 1228. Adding abscisic acid as an agronomic tool to make ginger rhizomes spicier.
- Lev-Mirom, Y., Ashkenazy, N., Klymiuk, V. et al. (2026) Ancient grains illuminate the mosaic origin of domesticated wheat. Nature Plants. https://doi.org/10.1038/s41477-026-02283-y This new paper offers a fascinating new take on the dynamics of wheat domestication.
- Garnier, E., et al. (2026) Leaf longevity and structure, fruit mass and phenology in 52 cultivated varieties and wild accessions of olive. Functional Ecology 40: 1193-1206. Leaf and fruit traits in wild and cultivated olive (Olea europaea).
- Scheer, K., et al. (2026) Rapid adaptive increase of amylase gene copy number in Indigenous Andeans. Nature Communications 17: 3822. Potato domestication likely increased salivary amylase in Andean peoples. See the commentary in Nautilus magazine.
- Smells like spring spirit. Nat. Plants 12, 669 (2026). The good folks at Nature Plants remind us that many of our sensory pleasures are gifts from plants.
- There are several recent interspecific analyses of fruit form and function and seed dispersal:
- Silveira, F. A. O., et al. (2026). Fruit traits: the ugly duckling in the whole spectrum of plant form and function. New Phytologist 250: 2001-2006. Making sense of fruit characteristics across species.
- Messeder, J. V. S., and T. A. Carlo. (2026) Fruit-quality tradeoffs generate asymmetry in plant reliance on mutualistic frugivores. Oikos e11848. The authors explore theories about fruit traits, seed dispersal, and frugivores using data from three wild Solanum species, relatives of the numerous Solanum species utilized by people.
- Jin, L., et al. (2026) Evolutionary history and the global distribution of seed dispersal modes in angiosperms. New Phytologist 250: 1217-1230. Seed dispersal is a key driver of seed and fruit characteristics. It is a large part of the story of which species become seed and fruit foods for humans, and what those foods are like.
- Two articles on the biology of black pepper (Piper nigrum):
- Santana, S., et al. (2026) The ecology of attraction: Fruit traits and frugivore diversity in neotropical Piper. Functional Ecology 40: 611-623.
- Metschina, D., et al. (2026) Population genetics and phylogenomic insights into the origin of economically important black pepper (Piper nigrum). American J. Botany 113: e70187
- E.W. Parkinson (2026) Fifteen thousand years of bioarchaeological data reveal life history trade-offs among Europe’s first farmers, Proc. Natl. Acad. Sci. U.S.A. 123 (17) e2505519123. Lots of research has shown that when people in Europe started farming, they got short and sick. This paper brings together a great deal of data to add some details to the picture. Basically, people were busy making babies instead of growing tall.
- Ren, S., et al. (2026). Plant flavonoids: Biosynthesis, regulation, and roles in biotic and abiotic stresses. Plant, Cell & Environment: e70540. Chemists and ecologists unite! This paper is a deeeeeep dive into the biochemistry and function of flavonoids, which help plants manage the slings and arrows of their sessile existence.
- Damián-Parizaca, A., et al. (2026). Dissentangling the Vine: Phylogenomics and Historical Biogeography of Vanilla (Orchidaceae). Preprint. There are more than 100 species in the pantropical genus Vanilla, but species in only one subgenus produces vanillin, the main aromatic component in commercial vanilla (from Vanilla planifolia). The authors clarify the closest wild relatives to Vanilla planifolia. This paper is timely for this month’s newsletter.
- Mićanović, N., et al. (2026) Underutilized Medlar (Mespilus germanica L.) Fruit: Polyphenol Extraction Optimization, Chemical Profiling, and In Vitro Pharmacological Evaluation. Plants 15: 1169. New research on an old fruit with very funky ripening biology.
- Oldie but Goodie: Raven PH. (2020) Plants make our existence possible. Plants People Planet 3:2–6. Peter Raven, one of the most important and globally influential botanists of the 20th century, died on April 25th at age 89. The paper we share here is not old, but it is definitely a good way to remember Raven’s influence. Peter Raven came of age in California but became the face of the Missouri Botanical Garden, having transformed it from a modest regional botanical garden to a major center for research and conservation. Raven literally wrote the textbook that, for the last 50 years, has introduced millions of students to botany. It has gone through numerous editions with various co-authors over the years, and we have several of these on our own shelves. Please see memorial commentary from Nautilus magazine and Mongaby.
References
1. A. T. Moles, D. D. Ackerly, C. O. Webb, J. C. Tweddle, J. B. Dickie, M. Westoby, A Brief History of Seed Size. Science 307, 576–580 (2005).
2. S. Bellot, R. P. Bayton, T. L. P. Couvreur, S. Dodsworth, W. L. Eiserhardt, M. S. Guignard, H. W. Pritchard, L. Roberts, P. E. Toorop, W. J. Baker, On the origin of giant seeds: the macroevolution of the double coconut (Lodoicea maldivica) and its relatives (Borasseae, Arecaceae). New Phytol. 228, 1134–1148 (2020).
3. P. D. Atangana, M. Simo-Droissart, B. Sonké, V. Droissart, Seed morphology and its relationships with phylogeny, lifeform and distribution of African orchid species. Seed Sci. Res. 35, 246–257 (2025).
4. What Tiny Orchid Seeds Reveal About Their Evolution, Botany One (2026). https://www.botany.one/what-tiny-orchid-seeds-reveal-about-their-evolution/.
5. J. R. Pannell, Sex, sexes, sex roles, and gender in land plants. Am. J. Bot. 110, e16195 (2023).
6. A. T. Moles, M. Westoby, Seed size and plant strategy across the whole life cycle. Oikos 113, 91–105 (2006).
7. M. R. Leishman, I. J. Wright, A. T. Moles, M. Westoby, “The evolutionary ecology of seed size.” in Seeds: The Ecology of Regeneration in Plant Communities (2000; https://www.cabidigitallibrary.org/doi/abs/10.1079/9780851994321.0031)CABI Books, pp. 31–57.
8. N. Li, Y. Li, Maternal control of seed size in plants. J. Exp. Bot. 66, 1087–1097 (2015).
9. N. Li, R. Xu, Y. Li, Molecular Networks of Seed Size Control in Plants. Annu. Rev. Plant Biol. 70, 435–463 (2019).
10. A. Linkies, K. Graeber, C. Knight, G. Leubner-Metzger, The evolution of seeds. New Phytol. 186, 817–831 (2010).
11. H. C. Muller-Landau, The tolerance–fecundity trade-off and the maintenance of diversity in seed size. Proc. Natl. Acad. Sci. 107, 4242–4247 (2010).
12. A. T. Moles, D. D. Ackerly, C. O. Webb, J. C. Tweddle, J. B. Dickie, A. J. Pitman, M. Westoby, Factors that shape seed mass evolution. Proc. Natl. Acad. Sci. 102, 10540–10544 (2005).
13. D. Bonte, H. Van Dyck, J. M. Bullock, A. Coulon, M. Delgado, M. Gibbs, V. Lehouck, E. Matthysen, K. Mustin, M. Saastamoinen, N. Schtickzelle, V. M. Stevens, S. Vandewoestijne, M. Baguette, K. Barton, T. G. Benton, A. Chaput-Bardy, J. Clobert, C. Dytham, T. Hovestadt, C. M. Meier, S. C. F. Palmer, C. Turlure, J. M. J. Travis, Costs of dispersal. Biol. Rev. 87, 290–312 (2012).
14. J. Arditti, A. K. A. Ghani, Tansley Review No. 110. Numerical and physical properties of orchid seeds and their biological implications. New Phytol. 145, 367–421 (2000).
15. T. Fichant, A. Ledent, F. Collart, A. Vanderpoorten, Dispersal capacities of pollen, seeds and spores: insights from comparative analyses of spatial genetic structures in bryophytes and spermatophytes. Front. Plant Sci. 14 (2023).
16. Y. Hashimoto, S. Fukukawa, A. Kunishi, H. Suga, F. Richard, M. Sauve, M.-A. Selosse, Mycoheterotrophic germination of Pyrola asarifolia dust seeds reveals convergences with germination in orchids. New Phytol. 195, 620–630 (2012).
17. S. A. Levin, *Helene C. Muller-Landau, *Ran Nathan, *Jérôme Chave, The Ecology and Evolution of Seed Dispersal: A Theoretical Perspective. Annu. Rev. Ecol. Evol. Syst. 34, 575–604 (2003).
18. A. T. Moles, M. Westoby, Seedling survival and seed size: a synthesis of the literature. J. Ecol. 92, 372–383 (2004).
19. M. Bogdziewicz, M.-C. A. Acuña, R. Andrus, D. Ascoli, Y. Bergeron, D. Brveiller, T. Boivin, R. Bonal, T. Caignard, M. Cailleret, R. Calama, S. D. Calderon, J. J. Camarero, C.-H. Chang-Yang, J. Chave, F. Chianucci, N. L. Cleavitt, B. Courbaud, A. Cutini, T. Curt, A. J. Das, H. Davi, N. Delpierre, S. Delzon, M. Dietze, L. Dormont, W. Farfan-Rios, C. A. Gehring, G. S. Gilbert, G. Gratzer, C. H. Greenberg, A. Guignabert, Q. Guo, A. Hacket-Pain, A. Hampe, Q. Han, K. Hoshizaki, I. Ibanez, J. F. Johnstone, V. Journé, T. Kitzberger, J. M. H. Knops, G. Kunstler, R. Kobe, J. G. A. Lageard, J. M. LaMontagne, M. Ledwon, T. Leininger, J.-M. Limousin, J. A. Lutz, D. Macias, A. Marell, E. J. B. McIntire, E. Moran, R. Motta, J. A. Myers, T. A. Nagel, S. Naoe, M. Noguchi, M. Oguro, H. Kurokawa, J.-M. Ourcival, R. Parmenter, I. M. Perez-Ramos, L. Piechnik, T. Podgórski, J. Poulsen, T. Qiu, M. D. Redmond, C. D. Reid, K. C. Rodman, P. Šamonil, J. Holik, C. L. Scher, H. S. Van Marle, B. Seget, M. Shibata, S. Sharma, M. Silman, M. A. Steele, J. N. Straub, I.-F. Sun, S. Sutton, J. J. Swenson, P. A. Thomas, M. Uriarte, G. Vacchiano, T. T. Veblen, B. Wright, S. J. Wright, T. G. Whitham, K. Zhu, J. K. Zimmerman, M. Zywiec, J. S. Clark, Linking seed size and number to trait syndromes in trees. Glob. Ecol. Biogeogr. 32, 683–694 (2023).
20. M. R. Leishman, M. Westoby, The Role of Large Seed Size in Shaded Conditions - Experimental-Evidence. Funct. Ecol. 8, 205–214 (1994).
21. K. Maleki, F. Vandelook, A. Saatkamp, K. Maleki, S. Heshmati, E. Soltani, Global Patterns in the Evolutionary Relations Between Seed Mass and Germination Traits. Ecol. Evol. 15, e71543 (2025).
22. O. Eriksson, K. Kainulainen, The evolutionary ecology of dust seeds. Perspect. Plant Ecol. Evol. Syst. 13, 73–87 (2011).
23. S. Diantina, C. McGill, J. Millner, J. Nadarajan, H. W. Pritchard, A. Clavijo McCormick, Comparative Seed Morphology of Tropical and Temperate Orchid Species with Different Growth Habits. Plants 9, 161 (2020).
24. Y. Ogura-Tsujita, T. Yukawa, A. Kinoshita, Evolutionary histories and mycorrhizal associations of mycoheterotrophic plants dependent on saprotrophic fungi. J. Plant Res. 134, 19–41 (2021).
25. J. M. Baskin, C. C. Baskin, Germination and Seed/Embryo Size in Holoparasitic Flowering Plants with “Dust Seeds” and an Undifferentiated Embryo. Bot. Rev. 88, 1–49 (2022).
26. S. Oikonomidis, C. A. Thanos, Orchid embryo-to-seed (E:S) ratio as an indicator of germination behaviour and its ecological implications. Seed Sci. Res. 34, 218–227 (2024).
27. D.-K. Zhao, Z.-M. Mou, Y.-L. Ruan, Orchids acquire fungal carbon for seed germination: pathways and players. Trends Plant Sci. 29, 733–741 (2024).
28. Z. Zhao, Y. Wang, L. Yang, X. Qian, A. Yang, J. Liu, H. Jacquemyn, T. Li, X. Xing, Metabolic Shifts and Nutrient Transfer Patterns in Orchid Seeds During Symbiotic Germination. Plant Cell Environ. 48, 6406–6420 (2025).
29. Mycorrhizal fungi of Vanilla: diversity, specificity and effects on seed germination and plant growth: Mycologia: Vol 99 , No 4 - Get Access. Mycologia.
30. E. R. Pansarin, K. Suetsugu, Mammal-mediated seed dispersal in Vanilla: Its rewards and clues to the evolution of fleshy fruits in orchids. Ecology 103, e3701 (2022).
31. E. R. Pansarin, Monkey as seed dispersers of Neotropical Vanilla involves social learning. Plant Biol. 27, 584–591 (2025).
32. E. R. Pansarin, Epiphyte Vanilla relies on birds as long-distance seed dispersers. Plant Biol. 27, 1497–1504 (2025).
33. A. P. Karremans, C. Watteyn, D. Scaccabarozzi, O. A. Pérez-Escobar, D. Bogarín, Evolution of Seed Dispersal Modes in the Orchidaceae: Has the Vanilla Mystery Been Solved? Horticulturae 9, 1270 (2023).
34. A. Damián-Parizaca, O. Peréz-Escobar, A. P. Karremans, A. Antonelli, J. P. Janovec, N. Mitidieri-Rivera, O. J. Fitzpatrick, A. Barona, X. Wu, M. E. Engels, M. R. Miranda, W. de la Cruz, G. Carnevali, G. Salazar, E. Hágsater, M. C. R. Pappas, D. Coayla, I. Tamayo-Cen, R. Menchaca, E. Smidt, M. A. Lozano-Rodriguez, Y. Ruiz, L. Vélez, H. Garzón, L. Baquero, G. Iturralde, Á. J. Pérez, M. Jiménez, S. Oliva, K. Cameron, Dissetangling the Vine: Phylogenomics and Historical Biogeography of Vanilla (Orchidaceae). bioRxiv [Preprint] (2026). https://doi.org/10.64898/2026.04.07.716943.
35. C. Watteyn, B. Reubens, J. B. A. Bolaños, F. S. Campos, A. P. Silva, A. P. Karremans, B. Muys, Cultivation potential of Vanilla crop wild relatives in two contrasting land use systems. Eur. J. Agron. 149, 126890 (2023).
36. C.-H. Yeh, K.-Y. Chen, Y.-I. Lee, Asymbiotic germination of Vanilla planifolia in relation to the timing of seed collection and seed pretreatments. Bot. Stud. 62, 6 (2021).
37. H. C. Harries, Germination rate is the significant characteristic determining coconut palm diversity. AoB PLANTS 2012, pls045 (2012).
38. M. Foale, J. Biddle, A. Bazrafshan, S. Adkins, “Biology, Ecology, and Evolution of Coconut” in Coconut Biotechnology: Towards the Sustainability of the ‘Tree of Life,’ S. Adkins, M. Foale, R. Bourdeix, Q. Nguyen, J. Biddle, Eds. (Springer International Publishing, Cham, 2020; https://doi.org/10.1007/978-3-030-44988-9_2), pp. 17–27.
39. C. R. Clement, D. Zizumbo-Villarreal, C. H. Brown, R. G. Ward, A. Alves-Pereira, H. C. Harries, Coconuts in the Americas. Bot. Rev. 79, 342–370 (2013).
40. F. C. Beveridge, S. Kalaipandian, C. Yang, S. W. Adkins, Fruit Biology of Coconut (Cocos nucifera L.). Plants 11, 3293 (2022).